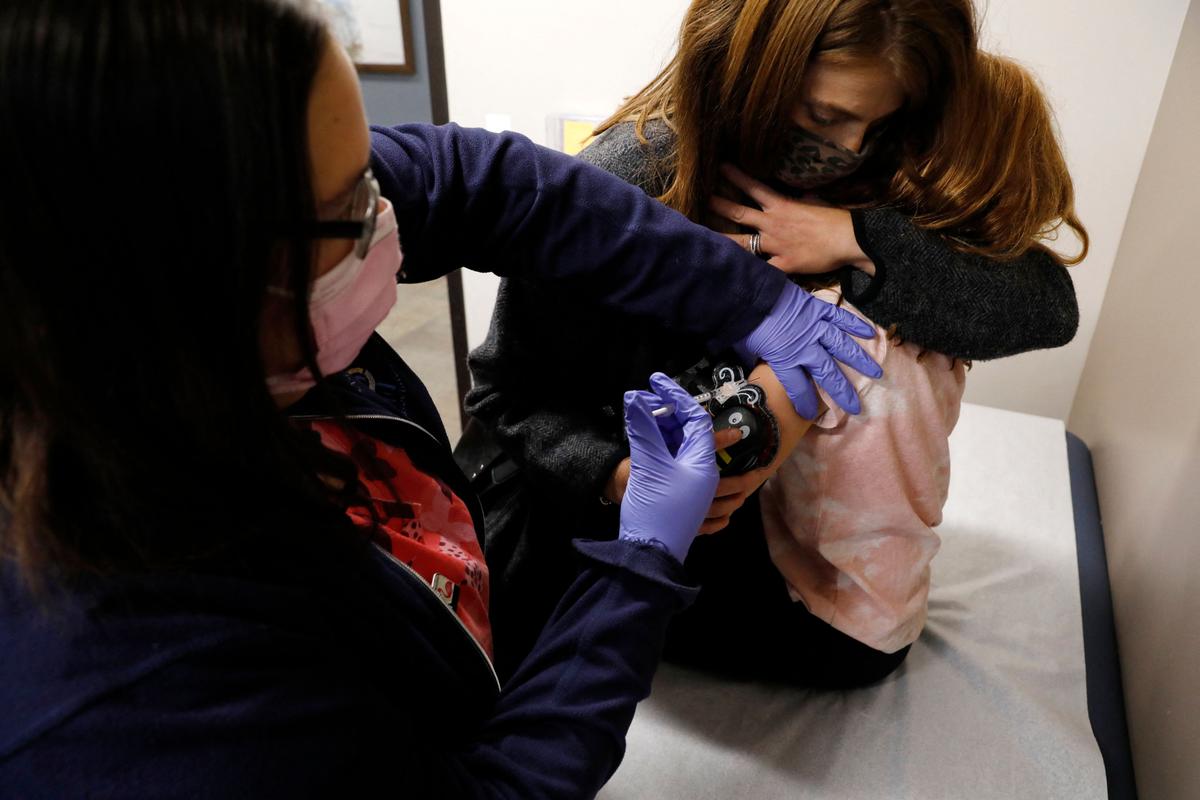
The FDA reported on Oct. 15 that it detected a safety signal for seizures/convulsions in children following COVID-19 vaccination.
U.S. drug regulators are refusing to say when they first identified a possible link between seizures and COVID-19 vaccines in children.
The paper went over results from safety monitoring on data from no later than April 2023.
An FDA spokeswoman declined to say, when asked, when regulators first detected the signal, continuing a pandemic pattern of withholding information regarding COVID-19 vaccines.
The study's corresponding author, an FDA statistician, did not respond to a request for comment.
Drug safety advocates said the FDA should be transparent, noting that convulsions are known complications of other shots, such as the measles and pertussis vaccines, and that they can cause brain inflammation.
"It is important for the FDA to be completely transparent about the safety signal suggesting there may be an increased risk for seizures in young children following COVID-19 shots," Barbara Loe Fisher, founder and co-president of the National Vaccine Information Center, told The Epoch Times via email.
"Unfortunately, there is a history of public health officials being reluctant to acknowledge serious vaccine reactions like convulsions for fear people will lose confidence in vaccine safety. But the public knows not everyone reacts the same way to vaccination, and trust is lost in public health policy recommendations when health officials either refuse or are slow to make information available that can help people weigh the risks for themselves when considering their own current health and medical history," she added.
Data From Paper
Researchers with the FDA and three health care companies, including CVS Health, analyzed data from health claims databases and found a signal for seizures/convulsions after COVID-19 vaccination in young children.They identified 72 cases of seizures/convulsions within seven days of a shot among the children, and compared rates with background rates from 2020.
The signal for seizures/convulsions for the young children “has not been previously reported for this age group in active surveillance studies of mRNA COVID-19 vaccines,” the researchers said, referring to the Pfizer and Moderna vaccines.
They said the signal “should be interpreted with caution and further investigated in a more robust epidemiological study," in part because when using background rates from 2022, the signal disappeared.
It's not clear whether the government plans to conduct an epidemiological study, and the FDA's delay in reporting the signal means other researchers haven't been able to act.
An FDA spokeswoman, in response to asking when the FDA identified the signal and whether it looked for the signal with more recent versions of the shots, said: "The FDA is confident in the safety, effectiveness and quality of the COVID-19 vaccines. The reviews conducted in these preprint papers are part of our ongoing safety surveillance efforts, which utilize a variety of data sources. The available data continue to demonstrate that the benefits of these vaccines outweigh their risks."
Pfizer and Moderna have not responded to requests for comment.
Seventy-five seizures and convulsions have been reported to the Vaccine Adverse Event Reporting System following COVID-19 vaccination among children under 6, according to the most recent data up to Sept. 29.
The system is co-managed by the FDA and the U.S. Centers for Disease Control and Prevention (CDC). The reports need verification, but often represent an undercount of the post-vaccination adverse events, studies have found.
Transparency Problems
The FDA and the CDC have repeatedly hidden information on COVID-19 vaccines, including signals.Defending its non-disclosure, the CDC said the results were "generally consistent" with those from data mining carried out by the FDA.
The FDA only started sharing documents it analyzed for its emergency authorization for the Pfizer and Moderna vaccines when ordered by federal judges.



No comments:
Post a Comment